|
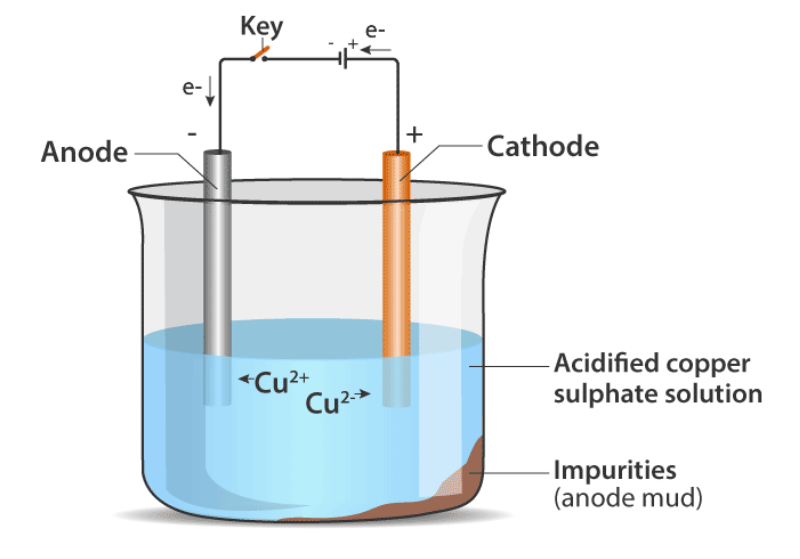
Carbon cannot reduce the oxides of sodium, magnesium, calcium, aluminium, etc., to the respective metals. This is because these metals have more affinity for oxygen than carbon. These metals are obtained by electrolytic reduction.
For example, sodium, magnesium and calcium are obtained by the electrolysis of their molten chlorides. The metals are deposited at the cathode (the negatively charged electrode), whereas, chlorine is liberated at the anode (the positively charged electrode). The reactions are : At cathode (Na⁺) + (e⁻) → Na ; At anode (2Cl⁻) → Cl2 + (2e⁻) Similarly, aluminium is obtained by the electrolytic reduction of aluminium oxide.
0 Comments
D is the midpoint of BC and E is the midpoint of AD in triangle ABC. prove that BE equal 3EM3/10/2023 In a triangle ABC, D is the midpoint of BC and E is the midpoint of AD. If BE is produced meets AC in M. Then prove that prove that BE = 3 EM construction: Draw DPIIAC Proof: BP=PM {by converse of of BPT) Now, Δ DEP ≅ ΔAEM PE = EM Now, BE = BP + PE = 2EM + EM = 3EM 10th similar triangle New pattern Question paper_2023-24 Download File 29(A) With the help of an activity, explain the action of saliva on the food we eat. |
Blog SeaRCH Link
All
Join Us For Update |